Over the past few years, OSD manufacturing has undergone a quiet revolution. While the dosage form itself remains unchanged, the processes, technologies, and strategies behind its production have advanced significantly. These innovations are helping manufacturers overcome long-standing challenges such as poor solubility, low bioavailability, and the safe handling of highly potent compounds.
For biotechs, these changes are more than technical upgrades, they represent opportunities to accelerate development timelines, improve patient outcomes, and reduce risk during scale-up.
1. Advanced Formulation Techniques
The rise of complex APIs has accelerated the need for innovation in formulation science. Many biotech molecules exhibit poor solubility or stability, making traditional approaches insufficient. New techniques are enabling better control over drug release, improved solubility, and enhanced patient outcomes.
Key advancements include:
- Spray Drying: Enhances particle uniformity and dissolution rates, critical for bioavailability.
- Nano-milling: Reduces particle size to the nanometre scale, increasing surface area and absorption.
- Multiparticulate Systems: Enables modified release profiles for complex dosing requirements.
These technologies are particularly valuable for biotech companies working with first-in-class or low-solubility molecules, where traditional formulation approaches may fall short. Selecting a CDMO with expertise in these techniques can significantly impact the success of early-phase development.
2. Potent Compound Handling
The increasing prevalence of oncology and targeted therapies has led to a rise in highly potent APIs (HPAPIs). Manufacturing these compounds safely requires specialised containment strategies and facility design.
Modern improvements include:
- Enhanced isolator technology for operator protection.
- Closed transfer systems to minimise exposure risk.
- Dedicated air handling and filtration for contamination control.
- Rigorous cleaning validation protocols to prevent cross-contamination.
However, not all facilities are equipped for the highest potency levels. Isolators are expensive, and industry-wide delays in isolator procurement are common. For biotechs developing highly hazardous materials, early consideration of containment capabilities is critical. Confirm whether your manufacturing partner has access to OEB4/OEB isolators and validated processes for HPAPI handling. This upfront diligence can prevent costly delays later in development.
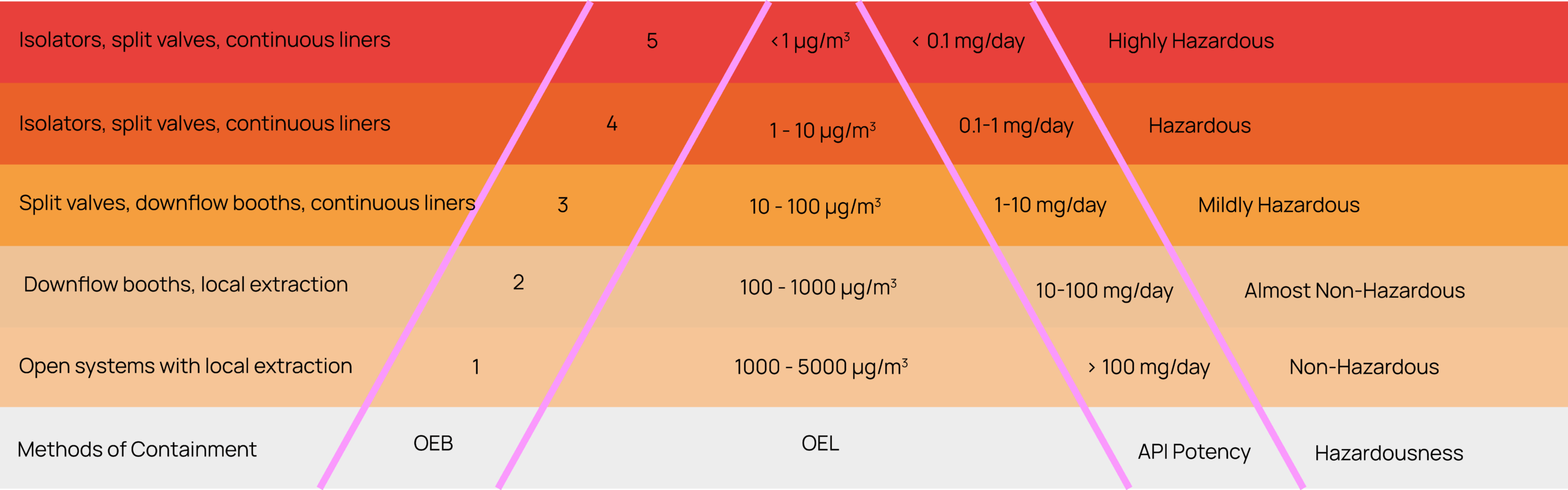
Figure 1: OEB Classifications
3. Modular and Flexible Manufacturing Suites
Flexibility is a hallmark of advanced OSD manufacturing. Facilities are increasingly designed with modular GMP suites that can be reconfigured based on project needs, dosage forms, or containment requirements.
This enables:
- Faster tech transfer between development stages.
- Parallel processing of multiple APIs, reducing bottlenecks.
- Reduced downtime between projects.
- Custom workflows tailored to specific molecule requirements.
For biotechs with diverse pipelines or evolving project scopes, this flexibility translates to faster timelines and reduced operational risk. At Upperton, our GMP suites are designed to accommodate these industry changes, offering reconfigurable spaces that adapt to project needs, whether for early-phase clinical supply or commercial-scale production.
4. Digitalisation and Process Analytics
Beyond physical infrastructure, digital transformation is reshaping OSD manufacturing. Advanced process analytical technology (PAT) and real-time monitoring tools are improving quality control and reducing variability.
Benefits include:
- Real-time data capture for critical process parameters.
- Predictive analytics to identify potential deviations before they occur.
- Automated reporting for regulatory compliance.
5. Regulatory Expectations and Global Compliance
As manufacturing technologies evolve, so do regulatory expectations. Agencies such as the FDA and EMA are placing greater emphasis on:
- Data integrity and traceability across the product lifecycle.
- Risk-based approaches to containment and cleaning validation.
- Continuous manufacturing as a pathway to efficiency and quality.
Biotechs should ensure their CDMO partner has robust quality systems and experience navigating global regulatory frameworks. Early alignment on compliance strategies can prevent delays during scale-up and commercialisation.
6. Scalability and Tech Transfer
One of the biggest challenges for biotechs is ensuring that early-phase solutions can scale effectively. Technologies like HME or spray drying may require specialised equipment and expertise at commercial scale. A CDMO with integrated development and manufacturing capabilities can streamline tech transfer, reducing risk and cost.
Tackling Poor Bioavailability with Early Formulation Strategies
What This Means for Biotechs
The evolution of OSD manufacturing offers significant advantages, but also introduces complexity. Biotechs must navigate:
- Technology selection: Which formulation approach best suits your molecule?
- Facility capabilities: Does your CDMO have the right containment and flexibility?
- Regulatory alignment: Are processes validated to meet global standards?
- Scalability: Can early-phase solutions transition smoothly to commercial production?
Early engagement with a CDMO that understands these dynamics is essential. At Upperton, we combine advanced formulation expertise, high-potency handling capabilities, and modular GMP suites to support biotechs from preclinical development through to commercial launch.